AI Compliance Shield for Clinical Trials
Protect your trials from invisible AI risks with Accelsiors’ ACCESHIELD™. A productized service ensuring EU AI Act compliance, data security, and multi-region alignment for emerging biotechs.

Welcome from Dr. Istvan Peter Horvath, Chief Legal & Risk Officer at Accelsiors
“I’m proud to introduce ACCESHILD™—our shield against AI risks, grounded in European expertise and designed to protect emerging biotechs like yours. As Accelsiors’ Chief Legal & Risk Officer, I’ve witnessed how ‘invisible AI’ threats, such as algorithmic drift or silent updates, can jeopardize clinical trials, especially in EU-inclusive studies. That’s why ACCESHILD™ offers a turnkey, execution-focused solution through three phases: Audit for mapping gaps, Remediation for fixing high-risk issues, and Governance for ongoing monitoring and controls. Leveraging our Switzerland-based insights and EU AI Act Readiness Playbook, it ensures defensible data under regulations like the AI Act, GCP, GDPR, and MDR/IVDR, while bridging to FDA ‘Context of Use’ for global alignment.
Beyond AI, we’re committed to NIS 2/ISO 27001-compliant data security, safeguarding healthcare infrastructure through robust risk management, incident reporting, and supply chain protections. Our audits definitively include NIS 2 compliance assessments, ensuring your trials are resilient against cyber threats and fully prepared for critical sector requirements.
We go beyond consulting by operationalizing compliance into your workflows, reducing risks by up to 50% and turning regulatory challenges into a competitive advantage, without you needing to build an internal AI or security team. Let’s secure your trials and accelerate your success together.”
ACCESHIELD™: Your Turnkey Defense Against AI Risks in Clinical Trials
If you’re an emerging US biotech navigating EU-inclusive studies, our specialized service line shields against ‘invisible AI’ threats like algorithmic drift, delivering operational compliance through Audit, Remediation, and Governance, without building internal teams.
Reduce AI risks by an estimated 50%
with our 2-week Readiness Sprint, turning regulatory burdens into a competitive edge.
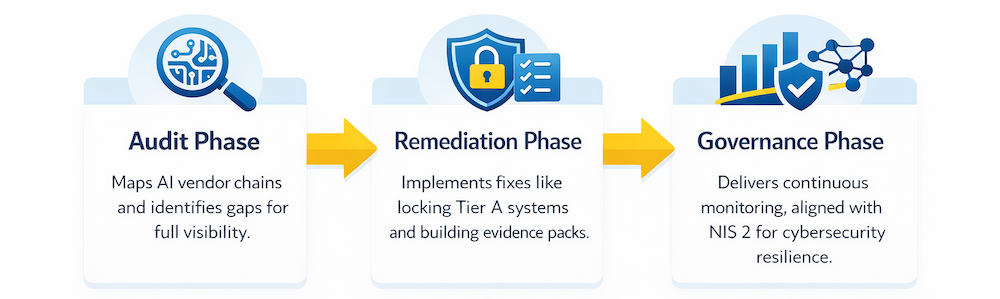
ACCESHIELD™ (AccelShield) is a productized service line addressing urgent ‘invisible AI’ risks for emerging US biotechs in EU-inclusive trials. Leveraging Switzerland-based expertise and our EU AI Act Readiness Playbook, it provides turnkey defense through three phases: Audit (mapping vendors and gaps), Remediation (fixing issues like silent updates), and Governance (ongoing monitoring and controls). This execution-focused product ensures data defensibility under EU AI Act, GCP, GDPR, MDR/IVDR, and bridges to FDA ‘Context of Use’ for global alignment—avoiding delays, scrutiny, and credibility issues.
Audit Phase: Maps AI vendor chains and identifies gaps for full visibility.
Remediation Phase: Implements fixes like locking Tier A systems and building evidence packs.
Governance Phase: Delivers continuous monitoring, aligned with NIS 2 for cybersecurity resilience.
Step 1: Audit Phase
Conduct a thorough mapping of AI tools and vendor chains to identify gaps, using our 3-Tier Risk Framework for quick prioritization.
Step 2: Remediation Phase
Address high-risk issues with practical fixes, such as the 2-week Readiness Sprint to secure systems and create inspectable evidence packs.
Step 3: Governance Phase
Establish ongoing monitoring and change controls, ensuring resilience against drift and alignment with evolving regs like the Digital Omnibus Regulation.
Benefits for Emerging Biotechs
Proactive Risk Mitigation: Slash AI threats by 50%, preventing mid-study delays and inspection issues.
Effortless Multi-Region Compliance: Bridge EU AI Act/GDPR to FDA standards without internal expertise.
Operational Edge: Turn burdens into advantages with tools like ‘once-only’ reporting readiness.

Frequently Asked Questions
A productized service shielding trials from AI risks via Audit, Remediation, and Governance phases.
It’s execution-focused, operationalizing compliance into workflows—not just high-level advice.
It bridges EU AI Act to FDA ‘Context of Use’ for seamless US-EU-Asia trials.
